113 of 115 enrolled patients underwent timely surgery; two had surgery delayed by more than two weeks
By Elana Gotkine HealthDay Reporter
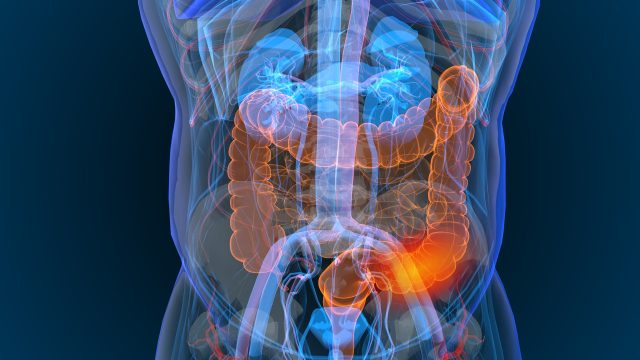
WEDNESDAY, June 5, 2024 (HealthDay News) — Neoadjuvant nivolumab plus ipilimumab has an acceptable safety profile in patients with locally advanced mismatch repair-deficient (dMMR) colon cancer, according to a study published in the June 6 issue of the New England Journal of Medicine.
Myriam Chalabi, M.D., Ph.D., from the Netherlands Cancer Institute in Amsterdam, and colleagues conducted a phase 2 study involving patients with nonmetastatic, locally advanced, previously untreated dMMR colon cancer treated with neoadjuvant nivolumab plus ipilimumab. Safety, defined by timely surgery, and three-year disease-free survival were the two primary end points.
The researchers found that 113 of the 115 enrolled patients underwent timely surgery; two had surgery delayed by more than two weeks. In five patients (4 percent), grade 3 or 4 immune-related adverse events occurred; no patients discontinued treatment due to adverse events. A pathological response occurred in 98 percent of the 111 patients included in the efficacy analysis, including 95 and 68 percent with a major pathological response and a pathological complete response, respectively (defined as ≤10 and 0 percent residual viable tumor). No patients had disease recurrence during a median follow-up of 26 months.
“The high proportion of patients with a pathological response observed after only four weeks of treatment in our study, together with the safety profile, may provide sufficient justification to provide immunotherapy to patients with radiographically assessed high-risk disease, especially if three-year disease-free survival data from this study are positive,” the authors write.
The study was funded by Bristol Myers Squibb, the manufacturer of nivolumab and ipilimumab.
Editorial (subscription or payment may be required)
Copyright © 2024 HealthDay. All rights reserved.